www.scifun.org |
||
 |
||
 |
||
 Photo credit: iStockphoto.com/drxy |
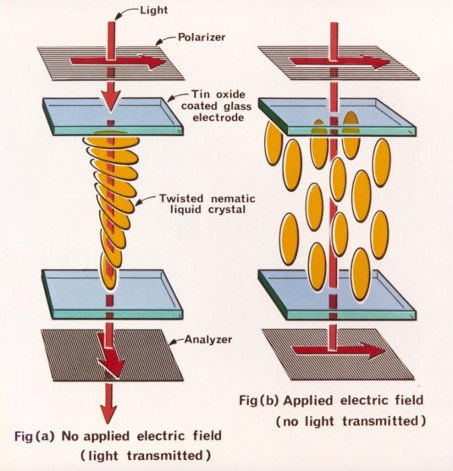
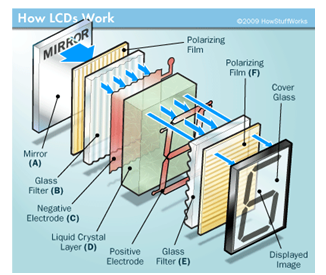
Have you ever wondered what lights up the numbers on your watch? Or pondered how the pictures form on your computer as you watch movies or play games? These are just a few everyday examples when you might be using items containing LCDs (liquid crystal displays). LCDs are common because they offer advantages over other display technologies previously used. They are thinner and lighter and draw much less power than cathode ray tubes (CRTs), which were used in the first television sets manufactured by Telefunken in Germany in 1934. |
But just what are these things called liquid crystals? The name "liquid crystal" sounds like a contradiction. We think of a crystal as a solid material like a diamond, usually hard as rock, and a liquid as a free-flowing material such as water. How could there be materials that exist in a state in between such different forms?
LCs are closer to liquids than solids as far as characteristics are concerned. It generally takes more heating to turn a solid into a liquid crystal than it does to turn that liquid crystal into a liquid. |
|